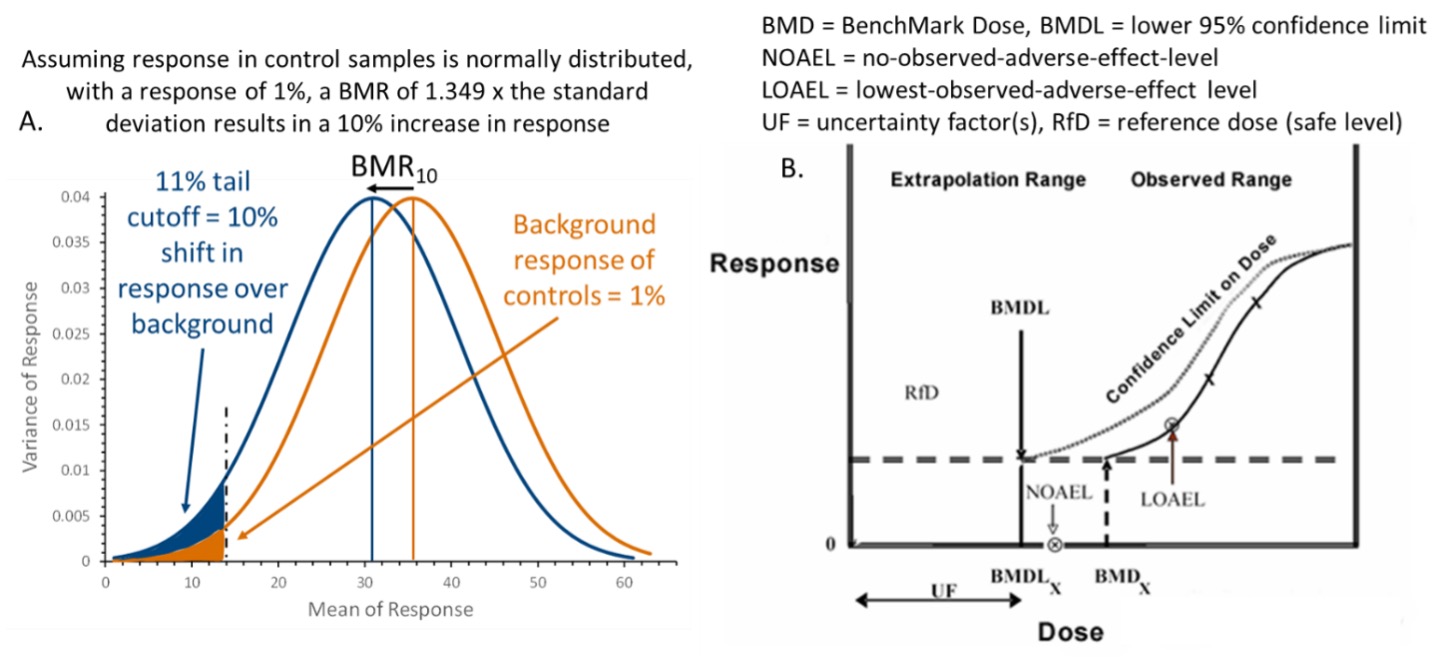
Toxicological approach to define the PDE for your cleaning validation process. - A3P - Pharmaceutical & Biotechnology Industry

Use of computational toxicology tools to predict in vivo endpoints associated with Mode of Action and the endocannabinoid system: A case study with chlorpyrifos, chlorpyrifos-oxon and Δ9Tetrahydrocannabinol - ScienceDirect

PDF) Derivation of Point of Departure (PoD) Estimates in Genetic Toxicology Studies and Their Potential Applications in Risk Assessment

A vape condensate collection method for degradant identification and toxicity screening - ScienceDirect

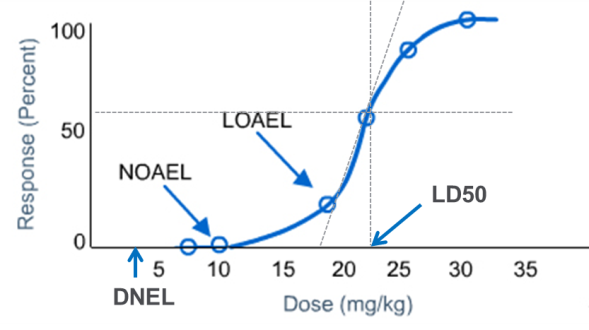
Point of departure (PoD) selection for the derivation of acceptable daily exposures (ADEs) for active pharmaceutical ingredients (APIs) - ScienceDirect

Stream episode Episode 4 - Toxicology and mental health with Nigel Brown & Jayanta Brahma by #IBMSChat Podcast podcast | Listen online for free on SoundCloud
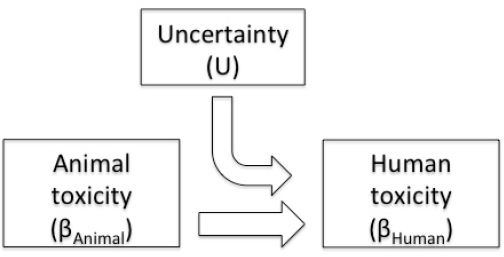
7 Derivation of Toxicity Values | Review of EPA's Integrated Risk Information System (IRIS) Process | The National Academies Press

Comprehensive interpretation of in vitro micronucleus test results for 292 chemicals: from hazard identification to risk assessment application | Archives of Toxicology

Artificial Intelligence-Based Toxicity Prediction of Environmental Chemicals: Future Directions for Chemical Management Applications | Environmental Science & Technology
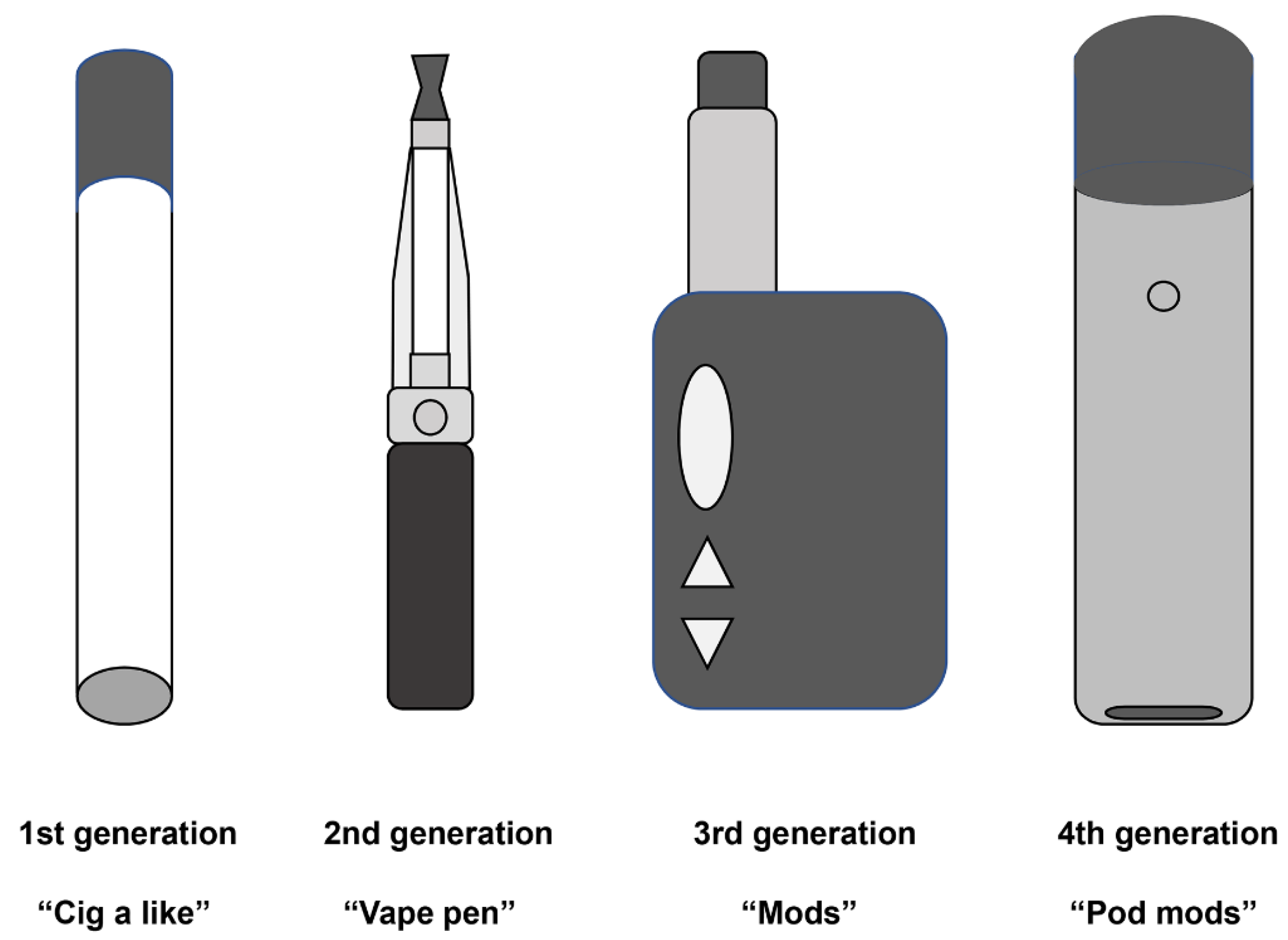
IJMS | Free Full-Text | A Review of Toxicity Mechanism Studies of Electronic Cigarettes on Respiratory System


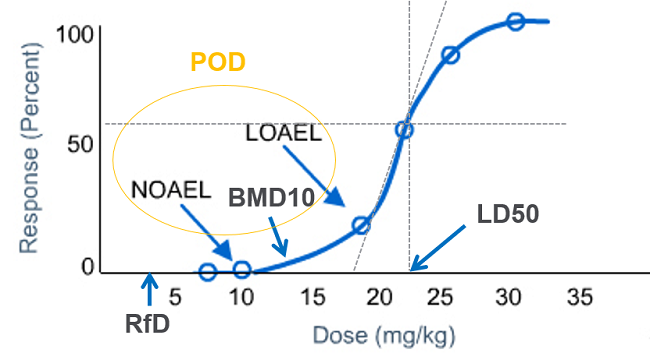
![PDF] Food for Thought … Systems Toxicology | Semantic Scholar PDF] Food for Thought … Systems Toxicology | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d96b72ff70c72dc6c6dc46ab31d54ef4700ce647/3-Figure1-1.png)